




Thiết bị
GeneProof Hepatitis B Virus (HBV) PCR Kit applies PCR technology to detect DNA of HBV virus in patient samples. The kit has CE-IVD certification for in vitro Diagnostic Use.
▶ Detection ability
▶ Technology
The HBV detection consists in amplification of a specific conservative DNA sequence of an open reading frame X (ORFx) and in measurement of fluorescence increase. The HBV presence is indicated by the FAM fluorophore fluorescence growth. The detection kit takes advantage of the ”hot start” technology, minimizing non-specific reactions and assuring maximum sensitivity.
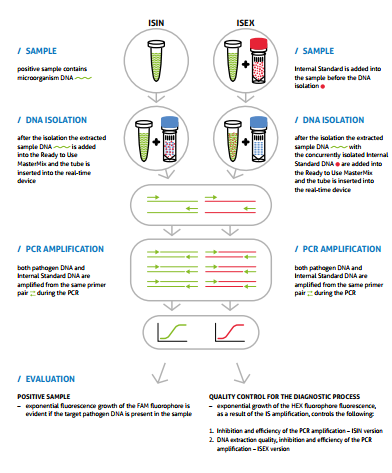

– Internal Standard (IS) is included in the PCR kit, controlling the possible inhibition of the PCR and the DNA extraction process quality (ISEX version
– Ready to Use MasterMix contains uracil DNA glycosylase (UDG), eliminating possible contamination of the PCR with amplification products
▶ Validated Specimen: plasma, serum
▶ Certification: CE-IVD for in vitro Diagnostic Use
▶ Order information
|
Product Name |
Cat. No |
Package |
|
GeneProof Hepatitis B Virus (HBV) PCR Kit |
HBV/ISEX/025 HBV/ISEX/050 HBV/ISEX/100 |
25 reactions 50 reactions 100 reactions |

TaqPath 1-Step Multiplex Master Mix (Không có ROX) là hỗn hợp RT-qPCR 4X, một ống, nhanh, nhạy, có thể lặp lại phát hiện lên đến bốn mục tiêu RNA/DNA khác nhau trong một phản ứng multiplex duy nhất. Đặc biệt hữu ích cho chẩn đoán và dược phẩm sinh học với các ứng dụng trong phát hiện mầm bệnh vi-rút và quy trình biểu hiện gen thông lượng cao, hỗn hợp bao gồm phiên mã ngược MMLV chịu nhiệt, dNTP, UNG và DNA polymerase Fast chịu nhiệt, tất cả trong một ống duy nhất. TaqPath được sản xuất theo hệ thống quản lý chất lượng ISO13485 mạnh mẽ và được thiết kế để tạo ra hiệu suất sản phẩm nhất quán sau từng lô.




Công cụ phòng lab tiên tiến nhất dành cho nghiên cứu khoa học.
Công nghệ giọt treo âm thanh Echo acoustic là bước đột phá cho giải pháp tự động hóa với khả năng không dính, không nhiễm mẫu, độ chính xác và tốc độ cao trong thao tác hút nhả mẫu lỏng sử dụng năng lượng âm thanh (Với mẫu từ 2.5 đến 25 nL).
Sóng âm thanh vận chuyển mẫu dưới dạng giọt treo với thể tích mẫu chính xác từ đĩa chứa cho tới đĩa đích treo. bên trên.
Với dải sản phẩm rộng bao gồm:
Echo 650 Liquid Handler
Echo 655 Liquid Handler
Echo 655T Liquid Handler
Echo 650T Liquid Handler
Echo 525 Liquid Handler
Echo 555 Liquid Handler
Optimize dependability and walk-away time. The Biomek i5 is the newest addition to our liquid handling portfolio representing the best of what has made Biomek an industry leading brand—combined with enhancements suggested by customers around the globe.
– Ideal for medium- to high-throughput workflows
– 25 deck positions
– 0.5 µL-1,000 µL (Multichannel) or 0.5 µL-5,000 µL (Span-8) pipetting volume ranges
– Single Multichannel head (96/384) or Span-8 pipetting with gripper
Tách chiết DNA từ tế bào và máu toàn phần:
– Dễ dàng tách chiết DNA từ máu toàn phần, buffy coat, mẫu tăm bông, hoặc mẫu nuôi cấy tế bào.
– Quy trình k cần sử dụng Proteinase K và các chất biến tính cho mẫu dịch sinh hoặc và mẫu nuôi cấy tế bào.


Clean-up PCR and other enzymatic reactions in as little as 2 minutes:
– Clean and concentrate up to 5 μg DNA with ≥ 6 μl elution in as little as 2 minutes with 0 μl wash residue carryover.
– Column design allows DNA to be eluted at high concentrations into minimal volumes of water or TE buffer.
– Eluted DNA is well suited for use in PCR, DNA sequencing, DNA ligation, endonuclease digestion, RNA transcription, radiolabeling, arrays, etc.


Rapid purification of DNA from cells and whole blood:
– Easy purification of high-quality DNA from whole blood, buffy coat, swabs, or cultured cells.
– Protocol excludes the use of Proteinase K and organic denaturants for biofluid and cell samples.™ are fracture resistant and chemically insert.
The Health Statistics shows that there are 200,000 tuberculosis patients and 30,000 deaths every year in Vietnam. The main cause of human tuberculosis is M. Tuberculosis, also called as tuberculosis bacteria.
GeneProof Mycobacterium tuberculosis PCR Kit applies PCR technology to detect DNA of Mycobacterium tuberculosis in samples. The kit has CE-IVD certification for in vitro Diagnostic Use.


GeneProof Hepatitis C Virus (HCV) PCR Kit applies PCR technology to detect RNA of HCV virus in patient samples. The kit has CE-IVD certification for in vitro Diagnostic Use.

TruSight Tumor 170 Kit includes library preparation reagent set and uses next-generation sequencing (NGS) technology to provide a comprehensive assessment of 170 genes that are commonly related to mutations in solid tumors and somatic variants.