APPLICATION
– Detection 40 mutations on exons 2, 3, 4 of KRAS gene


Detection 40 mutations on exons 2, 3, 4 of KRAS gene
Code: PNAR-1006
Packing: 25 Pounds/set
Certificate: CE-IVD
APPLICATION
– Detection 40 mutations on exons 2, 3, 4 of KRAS gene
TECHNOLOGY
– Based on PNA-mediated Real-time PCR clamping technology using PNA probe segment that is absolutely specific to the target sequence
– Sensitivity: detectable when mutation concentration <1%
INPUT FORM
– Input sample source: surgical tissue, FFPE tissue . sample
COMPATIBILITY SYSTEM
Compatible RT-PCR systems: Bior-ad CFX 96, Light cycler 480, ABI 7500/7900, StepOnePlus, Rotor-Gene Q
Panagene
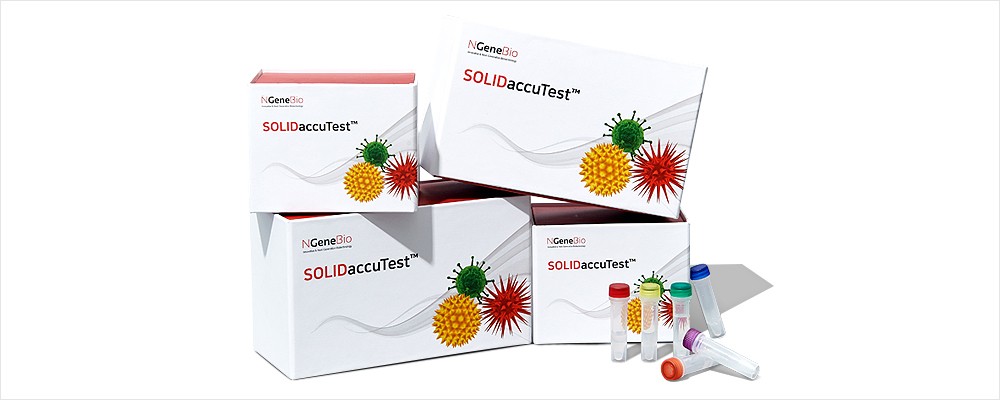


SOLIDaccuTest™ is an excellent tool for discovering variants associated with solid tumors using a comprehensive approach to next-generation sequencing (NGS). It reflects the latest research trends and is optimized for medical purposes by selecting essential genes of solid tumors including lung, colon, breast, skin brain, stomach and ovarian cancers, etc.



SOLIDaccuTest™ is an excellent tool to explorer variants associated with solid tumors using a comprehensive method of next-generation sequencing (NGS). It reflects the latest research trends and is optimized for the medical purpose by selecting essential genes of solid tumors including lung, colon, breast, skin brain, gastric, ovarian cancers, etc.


Model: NovaSeq 6000
Manufacturer: Illumina/USA
– Scalable platform
Match data output, time to results, and price per sample to study needs
– Flexible performance
Configure sequencing method, flow cell type, and read length to support a broad range of applications
– Streamlined operation
Increase lab efficiency with a simplified workflow and reduced hands-on time


The NextSeq 2000 Sequencing System uses patterned flow cells similar to those that power the NovaSeq™ 6000 System. The result is a highly flexible and scalable benchtop system that offers the highest cluster density flow cell of any on-market NGS system to date, driving down the cost per gigabase (Gb) of the sequencing run.


Model: NextSeq 1000
Manufacturer: Illumina
Origin: Singapore/United States
Launching year: 01/2020
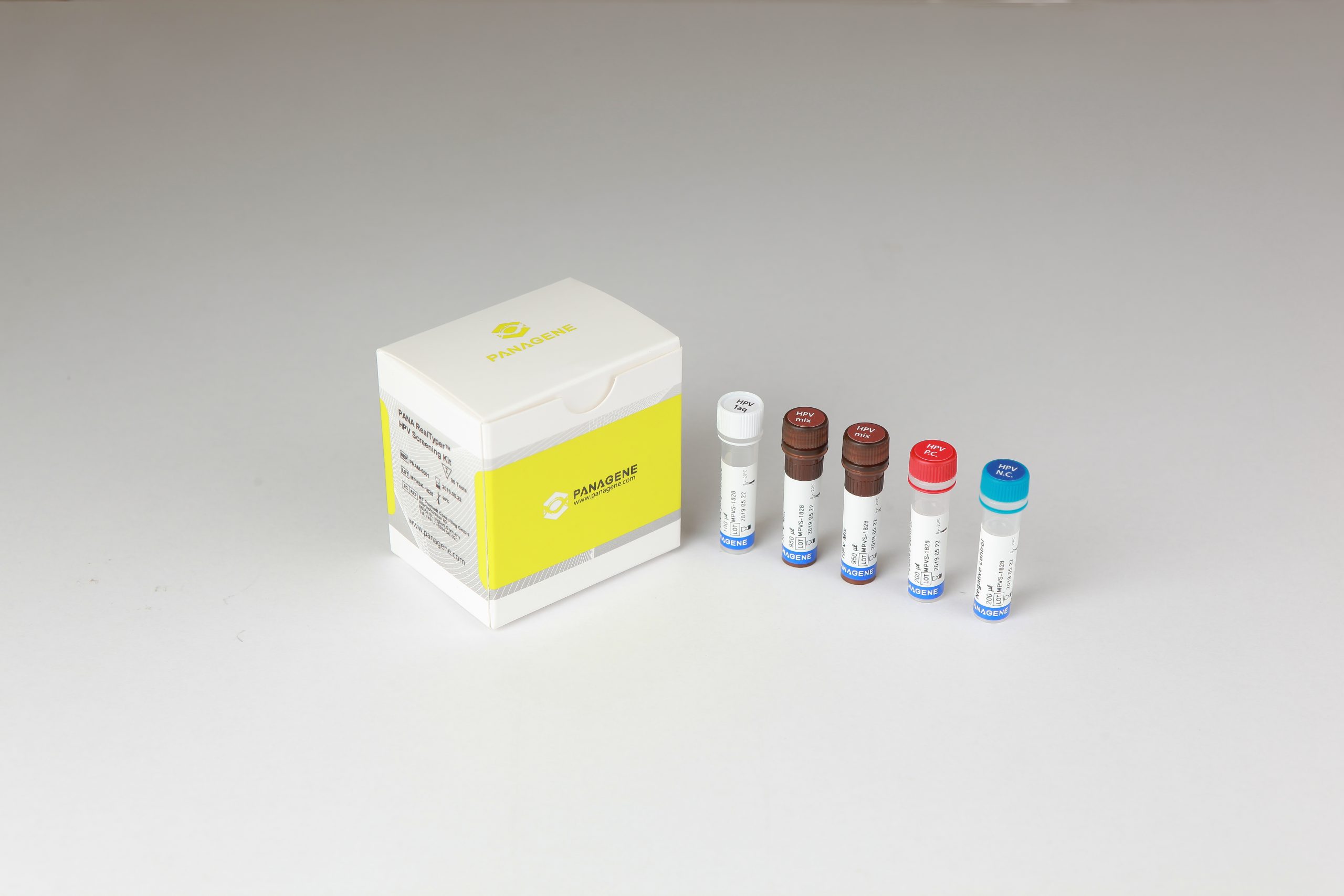

The PANA RealTyper™ HPV Screening Kit is a CE marked diagnostic device in accordance with the European Union in vitro Diagnostic Medical Device Directive 98/79/EC


DxFLEX is a new clinical flow cytometry platform derived from the successful CytoFLEX. The advanced sensitivity and intuitive software DxFLEX makes flow cytometry routine for both novice and expert flow cytometry technicians and promotes standardization. Functionality in the autoloader facilitates accurate results and sample tracking.


Model: CytoFLEX
Manufacturer: Beckman Coulter Life Sciences/ USA
The CytoFLEX Flow Cytometer is the most innovative flow cytometer system of Beckman Coulter. With compact design and high efficiency in work, CytoFlex provides and easy to use system with the impressive performance you need. Our system allows the researchers just focus on the science with no worry about instrument. Its superior sensitivity and resolution throughout all configurations give it the edge over other cytometry systems four times its size.
– Up to 3 lasers Violet-Blue-Red (V-B-R) and fully activated with 13 colors
– Ability to measure 100 nm particles with violet laser
– Includes 13 band pass filters: 450/45, 525/40 (2), 585/42, 610/20 (2), 660/10 (2), 690/50, 712/25, 780/60 (3)
– For higher throughput applications an optional plate loader module is available
– Smart software in acquisition and analysis CytExpert


Model: NovaSeq 6000
Manufacturer: Illumina/USA
– Scalable platform
Match data output, time to results, and price per sample to study needs
– Flexible performance
Configure sequencing method, flow cell type, and read length to support a broad range of applications
– Streamlined operation
Increase lab efficiency with a simplified workflow and reduced hands-on time


Model: iSeq 100
Manufacturer: Illumina/USA
Illumina’s smallest next-generation DNA sequencing system, with small, fast and efficient sequencing throughput, suitable for all labs
– Fast data generation: Suitable for small projects, on a dedicated device, low throughput with fast turnaround times
– Convenient operation: Control the sequencing process from start to finish and ensure independent sequencing instead of outsourcing.